Those stereoisomers which are differing in its configuration at only one chiral carbon atom are called as Epimers. The forward reaction is favored over the.

How To Determine The Meso Isomer In Bond Line Structures Chemistry Lessons Chemistry Organic Chemistry Study
We use these terms chiefly in organic chemistry.

. The number of possible structural arrangements for an asymmetrical molecule depends on the number of centres of asymmetry. Chiral centers that can be interchanged by rotation of a single bond. Draw the following molecules in two difference configurations about the double bond.
The division of a sample of a substance into progressively smaller parts produces no change in either its composition or its chemical properties until parts consisting of single. One of their most interesting type of isomer is the mirror-image stereoisomers a non-superimposable set of two molecules that are mirror image of one another. Interactions between particles in different molecules.
The existance of these molecules are determined by concept known as chirality. The particles cannot overlap necessitating a repulsive potential and they attract due to an dispersive induced-dipole-induced-dipole interaction. Molecules and elements differ in their chemical configuration.
Molecules that differ in configuration CANNOT be. 21 Organic molecules with only hydrogens and five carbon atoms cannot _____. They have different melting points and boiling points and different densities.
To convert between diastereomers only one chiral carbon atom must change configuration. 3 molecules of pentose 6 molecules of NADPH and 3 molecules of CO2 100 Which of the following describes the relationship between the straight chain forms of D-glucose and D-galactose given that they are non-cyclic molecules that differ only in stereochemical configuration at the fourth carbon. Chiral centers that are mirror images of each other.
Chiral centers that can be interchanged by rotation of a single bond. 4 Enantiomers and Diastereomers. Stereoisomers are isomers that differ in spatial arrangement of atoms rather than order of atomic connectivity.
There is no plane or angle for symmetry to establish. And if a molecule has chiral centers but as a whole is achiral due to the presence of a. Enantiomer is a term used in chemistry for mirror images of molecules that cannot be put over one another to give a same molecule.
Explicitly the total short-range interaction is often expressed as the sum of pairwise interactions between atoms located on different. Molecules such as the isomers of glyceraldehydethe atoms of which can have different structural arrangementsare known as asymmetrical molecules. This is not true for 8G.
Chirality cannot be localized around one atom or group of atoms. Chiral centers that are mirror images of each other d. Another example is the aggregation of M simple solute molecules to form a compact droplet shown in Fig.
Both terms conformation and configuration describes the spatial arrangement of a particular molecule. The key difference between conformation and configuration is that the conformations of the same molecule rapidly interconvert whereas the configurations of the same molecule do not readily interconvert. A have a branching carbon skeleton B have different combinations of double bonds between carbon atoms C have different positions of double bonds between carbon atoms D form enantiomers.
Molecules that differ in configuration can be all of the following except. Stereocenters must be different for molecules to be enantiomers. Chiral molecules cannot be superimposed on their mirror image.
Chiral centers that can be interchanged by rotation of a single bond. At least one stereocenter must be different and at least one stereocenter must be the same for molecules to be diastereomers. Chiral centers that are mirror images of each other.
Ie for n any given number of centres of asymmetry 2n different isomers of a. Name the molecules and be sure to include the appropriate. Molecules that differ in configuration CANNOT be.
Chiral centers that can be interchanged by rotation of a single bond. Molecule a group of two or more atoms that form the smallest identifiable unit into which a pure substance can be divided and still retain the composition and chemical properties of that substance. If two diasteromers with 2 or more chiral centers differ only in the absolute configuration see Cahn-Ingold-Prelog convention of a chiral carbon they are said to be epimers.
Chiral molecules are asymmetric meaning there is no symmetry at all. Molecules that differ in configuration CANNOT be. On the other hand elements can form numerous types of bonds based on electron arrangement.
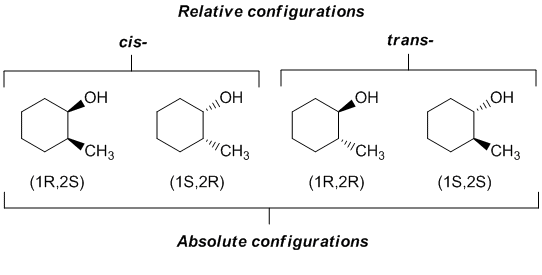
This function depends on the entire configuration of the solute molecule and cannot be split into pairwise additive indirect interactions. Cistrans configurational isomers are called Geometrical isomers in some textbooks. Which is the proper definition of equilibrium with respect to a biochemical reaction.
Chiral centers that are mirror images of each other. Molecules can form a specific type of bond ie covalent or ionic bonds. It is the property of the molecule as a whole.
Same IUPAC name for constitution different IUPAC prefix for configuration. Two molecules with the same constitution but different configuration ie. Molecules have atoms of similar or different elements whereas elements contain only similar atoms.
Biomolecules Introduction Structure and Functions- Carbohydrates. Configurational isomers cannot be interconverted without breaking bonds.


0 Comments